   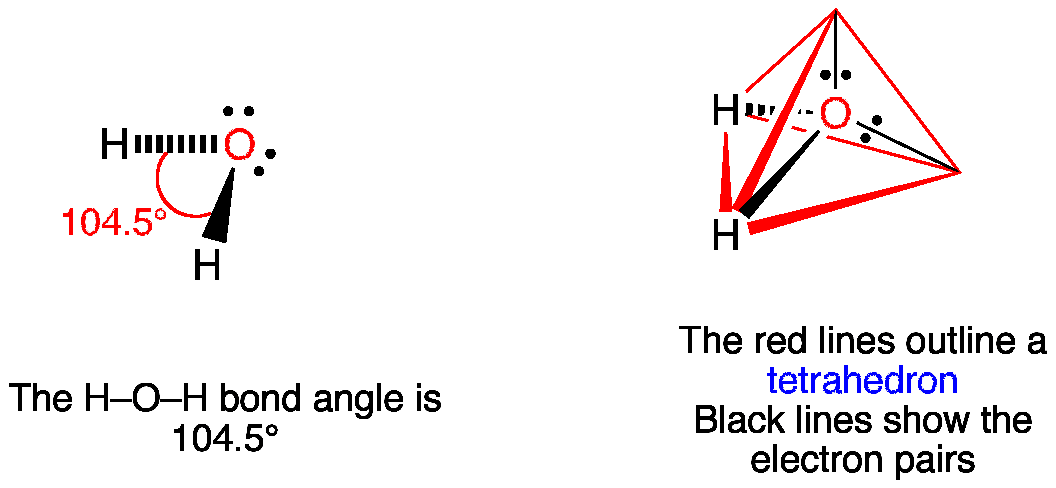
It is a set of rules used in chemistry to predict the geometry of a molecule. VSEPR theory stands for valence shell electron pair repulsion theory. Saturated Unsaturated and Supersaturated.Reaction Quotient and Le Chatelier's Principle.Prediction of Element Properties Based on Periodic Trends.The Lewis structures of many covalent molecules have been drawn and discussed in the last several sections of this chapter. Molecular Structures of Acids and Bases Learning Objective Predict the Valence Shell Electron Pair Repulsion, or VSEPR, shape of a covalent molecule, based on the bonds and lone pairs associated with the central atom of a Lewis structure.Ion and Atom Photoelectron Spectroscopy.Elemental Composition of Pure Substances.They really repel each other as they are only attracted to the oxygen atom in the water molecule. Application of Le Chatelier's Principle The reason water has a bent shape is that the two lone pair of electrons are on the same side of the molecule.Structure, Composition & Properties of Metals and Alloys.Intramolecular Force and Potential Energy.Variable Oxidation State of Transition Elements If there are only two bonds and one lone pair of electrons holding the place where a bond would be then the shape becomes bent.Transition Metal Ions in Aqueous Solution.Single and Double Replacement Reactions.Notes There are no stable AXE4, AX3E3, AX2E4 or AXE5 molecules. We are interested in only the electron densities or domains around atom A. Notice that #"SCl"_2# has a molecualr geometry that is very similar to water's, the only differences being the smaller bond angle (water has a bond angle of and the longer bond lenght (water has a bond lenght of #"95.84 pm"#. lone VSEPRTheory(MolecularShapes) the central atom, X an atom bonded toA, E alonepair on A pairs on X or other atoms, but we dont care. Repulsion between 2 lone pairs of electrons is greater than the repulsion between a bonding pair and a lone pair of electrons, the repulsion between 2. #"S"#, which is the molecule's central atom, has a steric number equal to 4 and a coordination number equal to 2.Įlectron geometry, which is determined by the steric number, will be tetrahedral, while molecular geometry, which is determined by the coordination number, will be bent. It is important to remember that Lewis structures are not meant to convey geometry, so it would be wrong to assume that the molecule is linear just by looking at this particular Lewis structure.Īll 20 valence electrons ( 6 from #"S"#, and 7 from each #"Cl"# atom) are accounted for by the Lewis structure, so VSEPR Theory can now be used to determine molecular shape. Start with the molecule's Lewis structure, which is drawn like this: Its a complicated acronym, but it means something thats not difficult to understand. #"SCl"_2# has a bent molecular geometry with bond angles of approximately and a bond lenght of #"201 pm"#. What is VSEPR VSEPR stands for Valence Shell Electron Pair Repulsion. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
